Inter-university Accelerator Centre, New Delhi, India
Heavy Ion Radiation Biology is an interdisciplinary applied science involving Atomic and Nuclear Physics, Molecular Biology and Biochemistry. The current research in this field investigates the effects of accelerated charged particles on the biological systems at the molecular level. In that sense, the research field that is intended to be pursued at IUAC may be termed Molecular Radiation Biology.
The Radiation Biology research facility in terms of a dedicated beamline and a molecular biology laboratory is very much suitable for carrying out fundamental research in this field keeping up pace with the other laboratories in the world. Various experiments can be designed to study the molecular mechanisms leading to the radiation-induced effects.
Various experiments may be planned by choosing suitable projectiles with appropriate energies to irradiate the samples of interest. The dedicated beamline for Heavy Ion Radiation Biology is designed for experiments using the low flux of accelerated particles. The irradiation is done using a computer-controlled system called ASPIRE. The support laboratory includes a cell culture facility and other equipment for sample preparation and post-irradiation treatments and analysis.
The obtained data may be used for :
- Modeling radiation effects
- Heavy Ion Cancer therapy
- Radiation protection in manned space flights
- Exploring different pathways to radiation induced effects
Research Programmes
- Effect of 45 MeV 7Li and 68 MeV 16O charged particles on the microsomal membrane fluidity. M. Srivastava, D. Choudhary, A. Sarma and R.K. Kale, Current Science, 74, No.1(1998) 58
- Effect of high LET radiation on biological membranes. D. Choudhary, M. Srivastava, A. Sarma and R.K. Kale, Radiat Environ Biophys, 37, No.3(1998), 177-185
- Response to glyoxalase system to low doses of mixed radiation. D. Choudhary, D.Chandra, S.P. Lochab, A. Sarma and R.K. Kale. Physica Medica, Vol XV, N.1, January - March 1999, page 27-33
- Effect of 7Li (45 MeV) ions on spinach leaves studied by thermoluminescence technique. Jyoti Gaikwad, S. Thomas, S. Kamble, P.B. Vidyasagar and A. Sarma, Nucl. Instr. And Meth. In Phys. Res. B 156 (1999) 231-235
- Effect of ionizing radiation: induced damage and protection in normal and denatured supercoiled DNA and minichromatins formed on them. S. De, S. Ray Chaudhuri, A. Sarma and A.R. Thakur, Indian J. Phys. 73B (5), 1999, 795 – 804
- Study of Carbonaceous Clusters in Irradiated Polycarbonate with UV-vis Spectroscopy, S. Gupta, D. Choudhary, A. Sarma, Journal of Polymer Science; Part B: Polymer Physics, Vol. 38, 2000, 1589 – 1594
- Effect of low doses of n-g mixed radiation on bioreductive enzymes in different tissues of mice. : D.Chandra, D.Choudhary, S.P.Lochab, A.Sarma, R.K.Kale, Physica Medica, Vol XVI, No.2, April-June 2000, page 83-87.
- Alpha-tocopherol protection against oxidative membrane damage induced by high LET radiation, Choudhary D., Sarma A, Kale RK, (2000), Research Communications in Biochemistry, Cell and molecular Biology, 4(3-4), 245-258.
- Irradiation of Thymine by 16O heavy ion in aerated condition. Pratyush Purkayastha, A.N. Talukdar and A. Sarma, Indian Journal of Pure and Applied Physics, Vol.39, November 2001, p 691-693
- Response of the FBX system to carbon beam: its potential as a dosimeter in heavy particle radiotherapy, MK Semwal, Milan Banerjee, Asiti Sarma and PB Vidyasagar, Phys. Med. Biol. 47 (2002), N179-N183
- Biological effects induced by swift heavy ions of Lithium on aqueous solution of plasmid pMTa4, J.O. Humtsoe, F.H.A. Schneeweiss, A. Srivastava, A. Sarma, R.N. Sharan, Radiation Effects and Defects in Solids, August 2003, Vol 158, p 603-607
- Proton and gamma ray induced gain degradation in bipolar transistor, S.R. Kulkarni, Asiti Sarma, G.R. Joshi, M. Ravindra and R. Damle, Radiation Effects and Defects in Solids, August 2003, Vol 158, p 647-654
- Optical absorption and photoluminescence studies on thin films and bulk crystals of LiF under swift heavy ion irradiation, F. Singh, A. Sarma, R.M. Montereali, F. Bonfigli, G. Baldacchini, D.K. Avasthi, Radiation Measurements, 2003, Vol 36, p 675-679
- Response of FBX Dosimeter to high LET 7Li and 12C ions, N. N. Bhat, D. Choudhary, A. Sarma, B.L. Gupta and K. Siddappa, : Radiation Physics and Chemistry, 68 (2003), 909-916
- Effect of Heavy Ion Irradiation on DNA DSB Repair in Methanosarcina Barkeri. Ray chaudhury S, Karmakar P, Choudhary D, Sarma A, Thakur A R, Anaerobe 2003 Feb;9(1):15-21
- Assessment of 1H heavy ion irradiation-induced effects in the development of rice (Oryza sativa L.) seedlings, M. Kalimullah, J.U. Gaikwad, S. Thomas, A. Sarma, P.B. Vidyasagar, Plant Science 165 (2003), 447-454
- Expression of NF-kappaB and ERK following heavy ion irradiation. Mitra AK, Sarma A, Krishna M, Verma NC, J Environ Pathol Toxicol Oncol 2004;23(1):53-9.
- Alteration in the expression of signaling parameters following carbon ion irradiation. Mitra AK, Bhat N, Sarma A, Krishna M, Mol Cell Biochem. 2005 Aug;276(1-2):169-73.
- Response to high LET radiation 12C (LET, 295 keV/microm) in M5 cells, a radioresistant cell strain derived from Chinese hamster V79 cells. Pathak R, Sarma A, Sengupta B, Dey SK, Khuda-Baksh AR, Int J Radiat Biol. 2007 Jan;83(1):53-63.
- Genotoxic effects in M5 cells and Chinese hamster V79 cells after exposure to 7Li-beam (LET=60 keV/microm) and correlation of their survival dynamics to nuclear damages and cell death. Pathak R, Sarma A, Dey SK, Khuda-Baksh AR Mutat Res. Genetic Toxicology and Environmental Mutagenesis 2007 Mar 30;628(1):56-66.
- Effect of 7Li radiation on the endogenous hormonal level on developing cotton fiber. Kunjal Bhatt, Asiti Sarma & Vrinda Thaker, Indian Journal of Experimental Biology Vol. 46, September 2008, pp. 677-680
- The influence of reduced glutathione on chromosome damage induced by X-rays or heavy ion beams of different LETs and on the interaction of DNA lesions induced by radiations and bleomycin. Geetanjali Pujari, A. Sarma, A. Chatterjee, Mutation Research, Genetic Toxicology and Environmental Mutagenesis 696 (2010) 154-159
- Activation of DNA damage response signaling in lung adenocarcinoma A549 cells following oxygen beam irradiation. Ghosh S, Narang H, Sarma A, Kaur H, Krishna M, Mutation Research, 2011 Aug 16;723(2):190-8. Epub 2011 May 14.
- DNA damage response signaling in lung adenocarcinoma A549 cells following gamma and carbon beam irradiation. Ghosh S, Narang H, Sarma A, Krishna M. Mutat Res. 2011 Nov 1; 716(1-2):10-9. Epub 2011 Aug 3.
- In vitro studies on radiosensitization effect of glucose capped gold nanoparticles in photon and ion irradiation of HeLa cells, Harminder Kaur, Geetanjali Pujari, Manoj K. Semwal, Asitikantha Sarma, Devesh Kumar Avasthi, Nuclear Instruments and Methods in Physics Research B 301 (2013) 7–11
- Study of in vitro toxicity of glucose capped gold nanoparticles in malignant and normal cell lines Harminder Kaur, Geetanjali Pujari, Asitikantha Sarma, Yogendra Kumar Mishra, Mi Kyung Jin, Bikesh K. Nirala, Nivedita K Gohil, Rainer Adelung, Devesh Kumar Avasthi, Advanced Materials Letters, Volume 4, Issue 12, Page 888-894
- Radiosensitivity and Induction of Apoptosis by High LET Carbon Ion Beam and Low LET Gamma Radiation: A Comparative Study. Ghorai A, Bhattacharyya NP, Sarma A, Ghosh U. Scientifica (Cairo). 2014;2014:438030. doi: 10.1155/2014/438030. Epub 2014 Jun 15
- Carbon ion beam triggers both caspase-dependent and caspase-independent pathways of apoptosis in HeLa and status of PARP-1 controls intensity of apoptosis. Ghorai A, Sarma A, Bhattacharyya NP, Ghosh U. Apoptosis. 2015 Apr;20(4):562-80. doi: 10.1007/s10495-015-1107-3.
- ASPIRE An automated sample positioning and irradiation system for radiation biology experiments at Inter-University Accelerator Centre, New Delhi, Ashok Kothari, P. Barua, M. Archunan, Kusum Rani, E.T. Subramanian, Geetanjali Pujari, Harminder Kaur, V.V.V. Satyanarayanan, Asitikantha Sarma, D.K. Avasthi, Radiation Measurements, 76, May 2015, Pages 17–22
- PARP-1 depletion in combination with carbon ion exposure significantly reduces MMPs activity and overall increases TIMPs expression in cultured HeLa cells, Ghorai A, Sarma A, Chowdhury P, Ghosh U., Radiation Oncology, 2016 Sep, 22;11(1), 126
- Targeting DNA repair with PNKP inhibition sensitizes radioresistant prostate cancer cells to high LET radiation, Srivastava P, Sarma A, Chaturvedi CM, PLoS One, 2018 Jan, 13(1): e0190516
- 12C-Beam Induces More Chromosomal Damage In Chemo-Radio-Resistant Cells than 16O-Beam, Utpal Ghosh, Regina Lichti Binz, Ratan Sadhukhan, Asitikantha Sarma, Subrata Kumar Dey, Martin Hauer-Jensen, Rupak Pathak, Research and Reviews: Journal of Pharmacy and Pharmaceutical Sciences, May 2018, Vol 7, Issue 2, p 9
- A12B4C3, an inhibitor of polynucleotide kinase/phosphatase enhances radio-sensitivity in PC-3cells exposed to carbon ion beam, Pallavi Srivastava, Asitikantha Sarma, Chandra Moini Chaturvedi, Journal of Innovations in Pharmaceutical and Biological Sciences (JIPBS), 2018, Vol 5(2), p27-31
- Reduction of metastatic potential by inhibiting EGFR/Akt/p38/ERK signaling pathway and epithelial-mesenchymal transition after carbon ion exposure is potentiated by PARP-1 inhibition in non-small-cell lung cancer, Chowdhury P, Dey P, Ghosh S, Sarma A, Ghosh U, BMC Cancer, 2019 Aug, 22;19(1):829
- Silymarin Mediates Apoptosis Through Activation of JNK/ERK Signaling Pathway in Human Colon Carcinoma Cells in Response to (60Co) Gamma Radiation, Mitu Lal, Saif Ahmad Khan and Damodar Gupta, Acta Scientific Cancer Biology 6.6 (2022) 01-13.
- Combined effects of carbon ion radiation and PARP inhibitor on non-small cell lung carcinoma cells: Insights into DNA repair pathways and cell death mechanisms, Payel Dey, Rima Das, Sandipan Chatterjee, Roni Paul, Utpal Ghosh, DNA Repair 144 (2024) 103778
Following research projects have been approved AUC approved research projects are being pursued:
- Exploring radio sensitizing potential of a natural phenolic compound in human lung carcinoma: highlighting macromolecular oxidation and DNA: Dr. Ujjal Das, UGC- DAE CSR, Kolkata Center.
- In vitro evaluation of carbon ion and gamma radiotherapy in combination with cisplatin in gallbladder carcinoma cell line G-415: Dr. Suresh Kumar Thokchom, Inter-University Accelerator Centre, Delhi.
- Radio-sensitisation of human cancer cell line with silver nano particle (SP): Dr. Sourav Ghosh, Inter-University Accelerator Centre, Delhi.
- In-Vitro Modulation of High LET Radiation-Induced DNA Damage and Repair by TLR agonist Mannan Oligosaccharide: Dr. Damodar Gupta, INMAS, DRDO, Delhi.
- Effect of Carbon Ion Radio-therapy in combination with GAS-STING agonist on triple negative breast cancer cell: Dr. Souren Paul, TERI School of Advanced Studies, Delhi.
- Investigating the Effects of Convolvulus prostatus (Shankhapushpi) Extracts on the Response of MCF-7, MCF-10, and MDA-MB-231 Cell Lines to High Energy Carbon-ion Irradiation: Dr. Saif Ahmad Khan, Inter University Accelerator Centre, Delhi.
- Impact of Lithium-Ion Beam Irradiation on Phenotypic Variation, Nutritional Composition and Biochemical Parameters in Glycine max (L.) Merr: Dr. Akshay Talukdar, IARI, Delhi.
- Investigating the ability of tardigrades to tolerate lethal ionizing radiations: Dr. Sandeep M Eswarappa, Indian Institute of Science, Bangalore.
- A Comparative Analysis of High LET Carbon Ion vs. Low LET Gamma Radiation- Induced Bystander Effects in Human NSCLC A549 Cells with PARP-1 Inhibitors: Mr. Roni Paul C/o. Dr. Utpal Ghosh, University of Kalyani.
- Exploring the impact of poly-ADP ribosylation on telomere maintenance in human lung cancer cells: Ms. Rima Das C/o. Dr. Utpal Ghosh, University of Kalyani.
- Modulation of cancer cell death by Anthocyanins: Mechanism based experimental approach: Ms. Zofa Shireen C/o. Prof. Sanjit Dey, University of Calcutta.
- in-Vitro Study on Radiation Sensitization of Nanoparticles in Photon and heavy ion Irradiation of Cancer Cells: Dr. Mudasir Ashraf Shah, Aligarh Muslim University, Aligarh.
- Transcriptomic and Proteomic Analysis of DNA Repair Genes of Human Prostrate Cancer Cell line exposed to high LET Radiation: Dr. Pallavi Srivastava, Galgotia University, Greater Noida.
- Study on utilizing Li-ion beam irradiation in Sorghum bicolor L. (Sorghum) to improve grain yield and abiotic stress tolerance, Ashok Badigannavar, BARC, Mumbai.
- Investigate the radiation induced bystander effect using a novel PDMS microwell array chips: Ms. Varshini Raju, Bharathiar University, Coimbatore.
Experimental Facilities
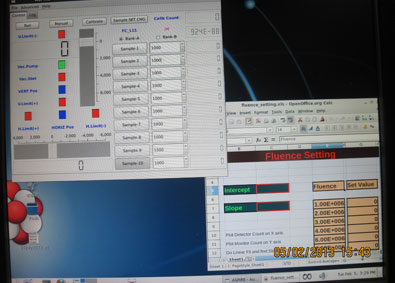
The research facility at IUAC provides a dedicated Radiation Biology Beam line equipped with the irradiation system called ASPIRE [Automatic Sample Positioning for Irradiation in Radiation Biology Experiments].
This is a computer controlled system that enables one to irradiate cells placed on 35 mm petri dishes kept in medium in a sterile condition one after another at quick succession with predetermined particle dose.
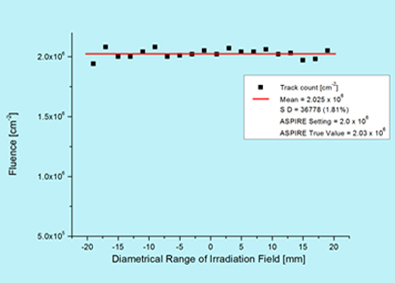
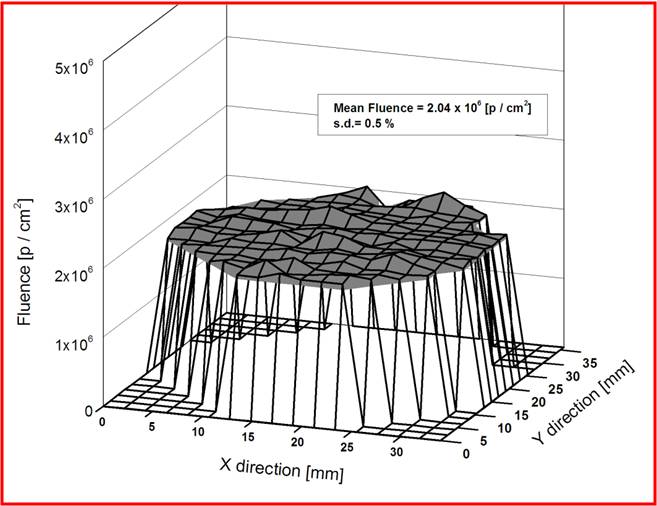
The uniformity as well as fluence verification has been done using irradiating SSNTD [CN-85] and subsequent etching. The tracks were counted using microscope over accurately measured areas across the entire irradiation field of 40 mm diameter. The uniformity over a field of 40 mm diameter is having 2 % standard deviation. The mean fluence is within 1 % of the electronically measured value at the centre of the field.
Photo Gallery
Radiation Biology Beam Line
Important information
| Beam | Beam energy from accelerator [MeV] | Energy on the Cell Surface [MeV] | Corresponding Entrance LET in water [KeV/µm] |
|---|---|---|---|
| 7 Li | 50 | 45 | 60 |
| 11 B | 65 | 48.7 | 218 |
| 12 C | 85 | 62 | 290 |
| 16 O | 100 | 56 | 614 |
All the above values give a tentative picture in order to plan an experiment. The parameters may be adjusted and evaluated during the beam time. The available beam energy also depends on the machine condition during the run.
Particle Flux : 1-2 x 105 particles/cm2 /sec
All the irradiation jobs are done at the atmospheric pressure. The samples have to be prepared in form of a monolayer. The attaching types of mammalian cells are grown on a 35 mm NUNC Petri-dish. The dishes are irradiated in a horizontal position. Non attaching cells can also be irradiated in the horizontal position using a special method. Irradiation with a pre-determined fluence and subsequent repetitions can be done using a the irradiation system called ASPIRE.

Group Members

Prof. Avinash Chandra Pandey
Director
[Programme Leader]
acpandey[at]iuac[dot]res[dot]in
Ext:215

Dr. Saif A. Khan
Scientist- G
[Lab in-charge]
Ext:333

Mrs. Supriya Mehta
Scientist-C
supriya[dot]mehta[at]iuac[dot]res[dot]in
Ext:225

Ms. Rushmika Rawat
Scientist- C
rushmika[dot]rawat[at]iuac[dot]res[dot]in
Ext:225

Mr. Abhay Sanjay Deore
Scientist C
abhay[dot]deore[at]iuac[dot]res[dot]in
Ext:225

Dr. Suresh Kumar Thokchom
Young Professional
suresh[dot]thokchom[at]iuac[dot]res[dot]in
Ext:343

Dr. Sourav Ghosh
Research Associate
sourav[dot]ghosh[at]iuac[dot]res[dot]in
Ext:343
Last Updated: 30 April 2026







_contrast_thumb.jpg)
![Coulter Cell Counter [Beckman Coulter],Countess image based Cell Coulter Cell Counter [Beckman Coulter],Countess image based Cell](https://iuac.res.in/themes/iuac/images/cellcount_thumb.jpg)
![CO2 Incubators, [NuAire] CO2 Incubators, [NuAire]](https://iuac.res.in/themes/iuac/images/incub_thumb.jpg)
![Bio safety Cabinet (Class II) [Esco] Bio safety Cabinet (Class II) [Esco]](https://iuac.res.in/themes/iuac/images/biosafe1_thumb.jpg)
![Bio safety Cabinet (Class II) [Heraeus] Bio safety Cabinet (Class II) [Heraeus]](https://iuac.res.in/themes/iuac/images/biosafe2_thumb.jpg)
![Gel Electrophoresis [Stratagene, Tarson], AFIGE [BIORAD], Semi Dry Blotter [BIORAD], PCR [Perkin Elmer] Gel Electrophoresis [Stratagene, Tarson], AFIGE [BIORAD], Semi Dry Blotter [BIORAD], PCR [Perkin Elmer]](https://iuac.res.in/themes/iuac/images/gel_thumb.jpg)
![UV Vis Spectrophotometer [Hitachi] UV Vis Spectrophotometer [Hitachi]](https://iuac.res.in/themes/iuac/images/UV_vis_thumb.jpg)
![PCR [Applied Biosystems] PCR [Applied Biosystems]](https://iuac.res.in/themes/iuac/images/pcr_thumb.jpg)
![96 Well Multi Mode Plate Reader [Perkin Elmer, Victor X5] 96 Well Multi Mode Plate Reader [Perkin Elmer, Victor X5]](https://iuac.res.in/themes/iuac/images/multimode_thumb.jpg)
![Centrifuge [Eppendorf] Centrifuge [Eppendorf]](https://iuac.res.in/themes/iuac/images/centri_thumb.jpg)
![Fluorescence Microscope [Zeiss] Fluorescence Microscope [Zeiss]](https://iuac.res.in/themes/iuac/images/zeiss_thumb.jpg)
